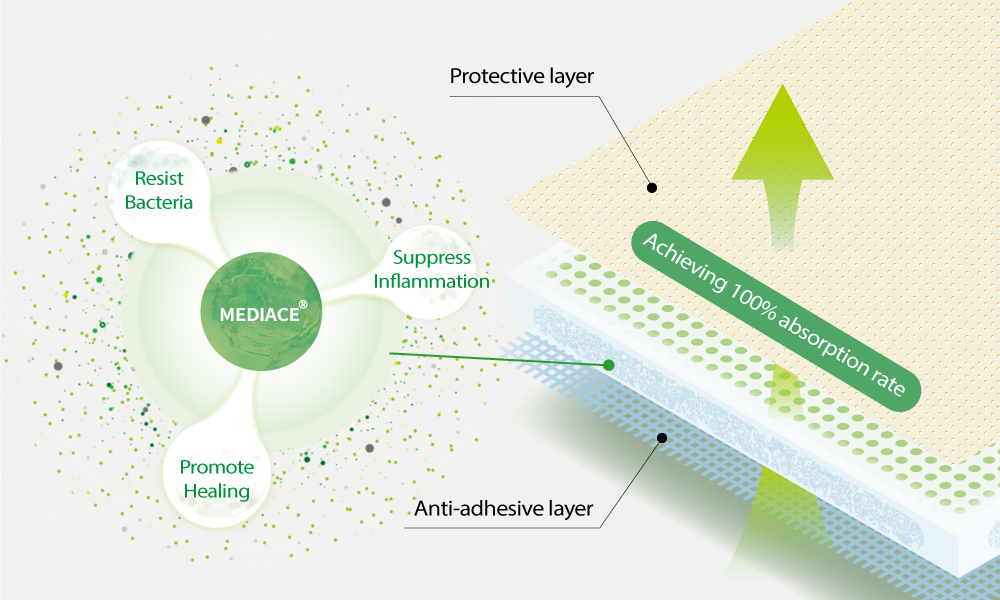
On January 18th, AMS BioteQ (TPE: 6864) announced the successful approval of its application for a novel invention patent in Taiwan titled “ARTIFICIAL DRESSING AND USE OF ARTIFICIAL DRESSING FOR PROMOTING WOUND” by the Intellectual Property Office of the Ministry of Economic Affairs, R.O.C. The company has combined the exclusive natural antimicrobial component, MEDIACE®, with artificial dressing to develop an groundbreaking antimicrobial medical material. This innovative solution caters to ailments including diabetic ulcers, bedsores, and burns, particularly in patients with prolonged bed rest. The dressing not only absorbs exudate but also releases active ingredients simultaneously, achieving both antimicrobial and therapeutic effects.”
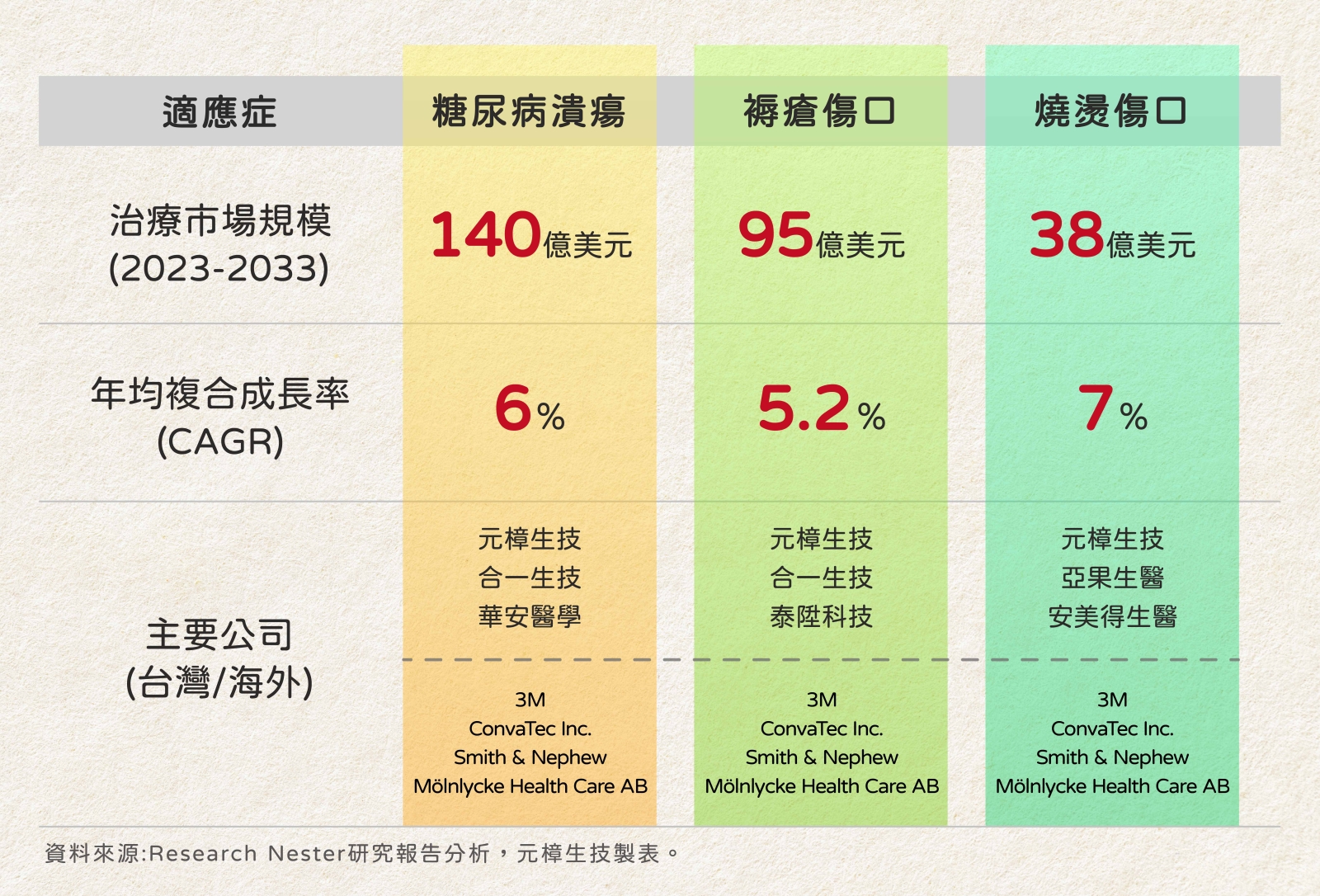
Based on the report analysis by Research Nester, the escalating prevalence of chronic wounds is anticipated to drive a surge in market demand. The projected market size for antimicrobial wound dressings is expected to exceed USD 5 billion by the end of 2036, exhibiting a growth rate of approximately 7% over the next decade. This trend poses financial challenges in the future wound care industry. Hence, there is a growing emphasis on antimicrobial dressings as a pivotal element in the market.
The Chairman of AMS BioteQ announced the integration of the innovative SIPSIP series with natural antimicrobial components, positioning it as a key development focus for the company. While most commercially available dressings adopt silver ions for antimicrobial effects, SIPSIP series may lead to minor allergic reactions and swelling in most wounds scenario. Striking a balance between accelerating wound healing and minimizing adverse reactions is crucial in the wound care industry. AMS BioteQ, through extensive preclinical testing and utilizing exclusive patented extraction technology ExtrO, alongside the unique physical structure of their proprietary dressings, has successfully developed an antimicrobial dressing featuring MEDIACE®. This breakthrough marks a new era in wound care, offering efficacy comparable to silver ions with lower side effects.
R&D Supervisor, Mr. Ying-Ting Yeh stated that AMS BioteQ advocates for 'Moist Treatment' in wound care. Animal and clinical trials have demonstrated the antimicrobial efficacy of their novel wound dressing, showcasing its ability to combat infections and reduce inflammation without any observed side effects. Significantly, the innovative wound dressing has demonstrated exceptional efficacy in diabetic mouse wound models, showcasing impressive recovery outcomes. With remarkable anti-adhesive properties, high absorbency, and tissue regeneration capabilities, it is uniquely tailored to meet the specialized wound care needs of the elderly and diabetic populations, positioning it as a highly competitive product in the market.
AMS BioteQ deploys the dual-track strategy in developing innovative wound care products, blending medical materials and pharmaceutical research. With approvals from TFDA and the U.S. FDA, the SIPSIP Foam has garnered international attention, leading to inquiries for collaboration from Japan, MENA, ASEAN, and beyond. The company focuses on gathering market data and expert feedback to strengthen international sales niches, positioning SIPSIP as a leading brand in the global medical market.